10/18/2021
High-Efficiency Production of Terpene Precursors for Next-Generation Biofuels
Optimizing the E. coli lepidopteran mevalonate pathway
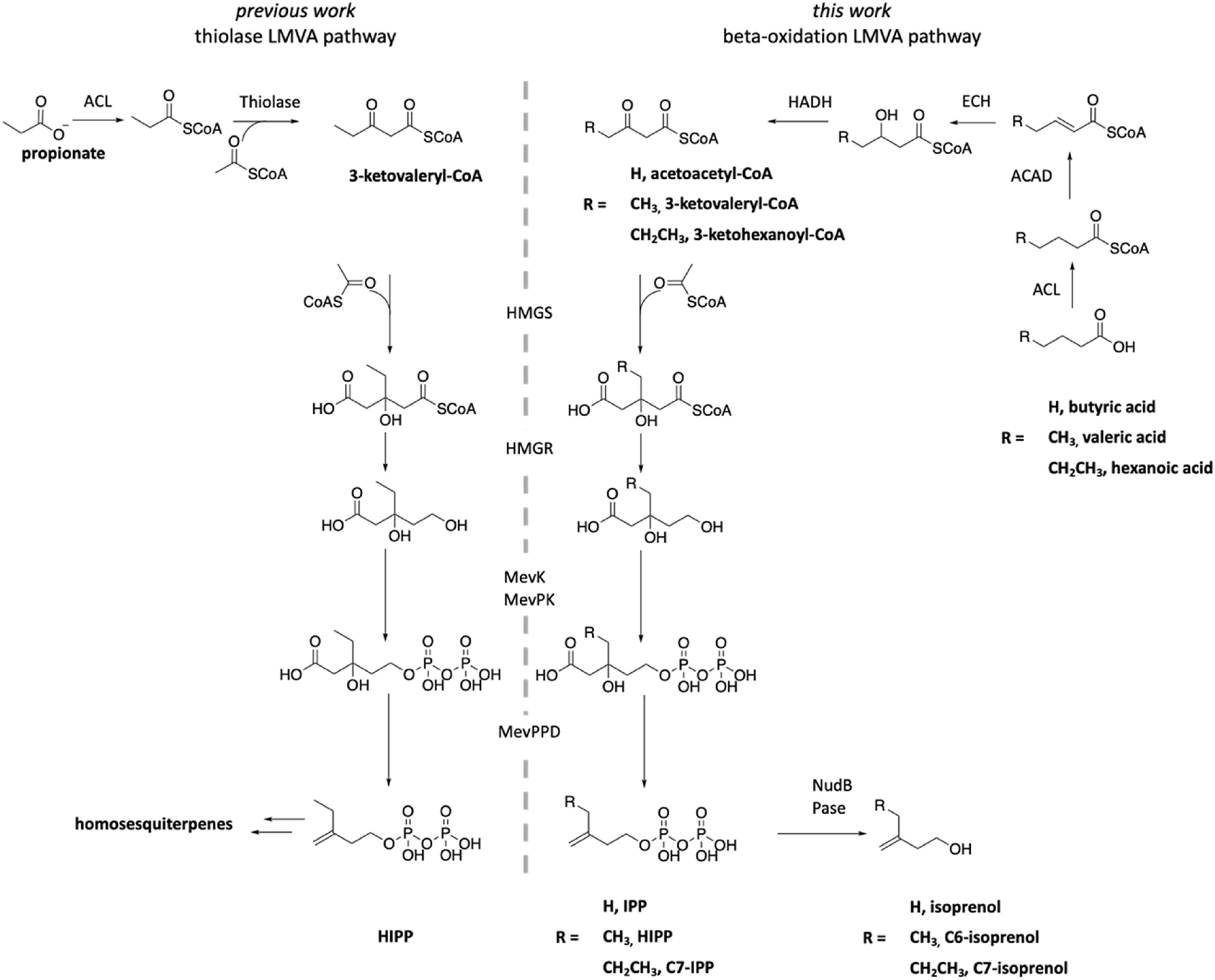
The natural lepidopteran mevalonate (LMVA) pathway (left) depends on a thiolase to produce the key intermediate, 3-ketovaleryl-CoA, while the new pathway (right) employs beta-oxidation enzymes to afford this intermediate and the analogs thereof, to increase the productivity of the whole pathway.
[Reprinted from Metabolic Engineering, Vol 68. B. Pangade et al. Lepidopteran mevalonate pathway optimization in Escherichia coli efficiently produces isoprenol analogs for next-generation biofuels. Pages 210-219, Copyright 2021, with permission from Elsevier.]
The Science
Terpenes constitute the largest class of natural products with over 55,000 compounds with versatile applications including drugs and biofuels. Introducing structural modifications to terpenes through metabolic engineering presents an efficient and sustainable way to improve their properties. The lepidopteran mevalonate (LMVA) pathway, an essential metabolic pathway in eukaryotes, archaea, and some bacteria, produces isopentenyl pyrophosphates (IPP) that serve as terpene building blocks. Scientists with the Joint BioEnergy Institute report optimization of the LMVA pathway in E. coli toward efficient production of six-carbon analogs of IPP as terpene precursors. The hydrolyzed products of these building block analogs are potential next-generation biofuels.
The Impact
The study not only optimizes the LMVA pathway, setting the basis for homoterpene biosynthesis to expand terpene chemical space, but also provides an efficient pathway to produce isoprenol analogs as next generation biofuels from sustainable feedstocks.
Summary
Pathway engineering involved first linking the LMVA pathway to NudB, a promiscuous phosphatase, resulting in production of C6-isoprenol, a six-carbon analog of isoprenol. The upstream portion of the LMVA pathway in E. coli was then redirected from a thiolase-dependent pathway to a beta-oxidation pathway. The beta-oxidation LMVA pathway transforms valeric acid, a platform chemical that can be produced from biomass, into C6-isoprenol at a titer of 110.3 mg/L. In contrast, the existing thiolase pathway uses propionic acid as a feedstock and produces only 5.5 mg/L C6-isoprenol. Knocking out the E. coli endogenous thiolase genes further improved the C6-isoprenol titer to 390 mg/L, implying efficient production. The beta-oxidation pathway also converts butanoic acid into isoprenol and hexanoic acid into isoprenol’s seven-carbon analog, C7-isoprenol, suggesting that the beta-oxidation LMVA pathway produces IPP and C7-IPP from the corresponding fatty acids. Fuel property tests revealed that the longer chain isoprenol analogs have lower water solubilities, similar or higher energy densities, and comparable research octane number boosting effects to isopentenols.
Principal Investigator
Jay Keasling
Lawrence Berkeley National Laboratory
jdkeasling@lbl.gov
BER Program Manager
Shing Kwok
U.S. Department of Energy, Biological and Environmental Research (SC-33)
Biological Systems Science Division
shing.kwok@science.doe.gov
Funding
This work was funded by the DOE Joint BioEnergy Institute (http://www.jbei.org), supported by the U.S. Department of Energy, Office of Science, Biological and Environmental Research Program, through Contract DE-AC02-05CH11231 between Lawrence Berkeley National Laboratory and the U.S. Department of Energy. This work was also funded by the Co-Optimization of Fuels & Engines (Co-Optima) project sponsored by the U.S. Department of Energy (DOE) Office of Energy Efficiency and Renewable Energy (EERE), Bioenergy Technologies and Vehicle Technologies Offices.
References
Pangade, B.; Li, J.; Eiben, C. B.; Oksen, E.; Barcelos, C.; Chen, R.; Englund, E.; Sundstrom, E.; Keasling, J. D. “Lepidopteran mevalonate pathway optimization in Escherichia coli efficiently produces isoprenol analogs for next-generation biofuels.” Metabolic Engineering, 68 (2021). [DOI: 10.1016/j.ymben.2021.10.007]
