Membrane Potential Dynamics during the Cell Cycle of Single Proliferating and Nonproliferating Bacterial Cells
Authors:
Kiran Bharadwaj1*(kiranbharadwaj@g.ucla.edu), Debjit Roy,1 Eduardo Charbon,2 Evan Miller,3 Xavier Michalet,1 Ted Laurence,4 and Shimon Weiss1
Institutions:
1University of California–Los Angeles; 2École Polytechnique Fédérale de Lausanne; 3University of California–Berkeley; and 4Lawrence Livermore National Laboratory
Goals
- To develop optical probes that use the quantum confined stark effect and quantum tunneling techniques to quantitatively investigate bacterial membrane potentials (MP).
- To develop advanced detectors for quantum technologies, including time-gated single-photon avalanche diode (SPAD) array imagers and time-correlated single- photon counting (TCSPC) avalanche diode (SPAD) arrays for various imaging
- To develop the phasor fluorescence lifetime imaging microscopy (phasor-FLIM) approach for classical pulsed laser light MP imaging using the SwissSPAD3 detector, and install current and future detectors on an inverted microscope with a scanning stage and a biofilm microfluidic
- To develop and benchmark non-classical light FLIM for membrane potential imaging using entangled two-photon absorption and antibunching g(2)(r) imaging of MP.
- To test both classical and non-classical light MP imaging techniques on DOE-relevant model biofilms of Bacillus subtilis and Shewanella oneidensis MR-1.
Abstract
Membrane potential (MP) is a crucial aspect of cellular physiology. Recent studies have explored its role in cell-cell communication and metabolic coordination within bacterial biofilms. MP plays an important role in broadcasting the metabolic status of cells located in different parts of the biofilm. MP has also been shown to play an important role in the stress response of microbial cells under various stressors.
Despite the expanding understanding of MP’s role in bacteria, little is known about its changes during the cell cycle as well as under different growth conditions. In this project, the team studies MP changes in single bacterial cells associated with different growth conditions (proliferating vs non-proliferating) and under various stressors, using VoltageFluor (VF) dyes. VF dyes insert into the lipid bilayer of the bacterial cell’s plasma membrane. They are presumed to exhibit electric field-dependent photo-induced electron transfer (PeT) process that modulates the fluorescence quantum yield and the excited state lifetime of the dye. Preliminary intensity-based widefield fluorescence measurements have been performed over long periods of time to record MP fluctuations in a single cell, in both rapidly growing as well as inhibited cell cultures of Bacillus subtilis (B. subtilis). Very different MP time-trajectories have been recorded for proliferating and non- proliferating cells (Figure 1).
Further experiments utilizing the dye’s excited-state lifetime as an MP readout are being performed using (1) a commercial confocal FLIM and (2) a home-built widefield FLIM set-up that utilizes a time-gated SPAD camera and a phasor-based analysis to quantitively capture small changes in resting MP during the cell cycle. A better understanding of MP changes in proliferating bacteria may provide better tools to manipulate microbial cell growth.
The team plans to image whole biofilms and resolve MP changes in individual cells within the community under steady-state conditions (i.e., steady-state fluctuations) and dynamically under non-equilibrium conditions upon introducing various environmental stressors, spatial gradients, nutrient limitation, and substrate consumption conditions.
Image
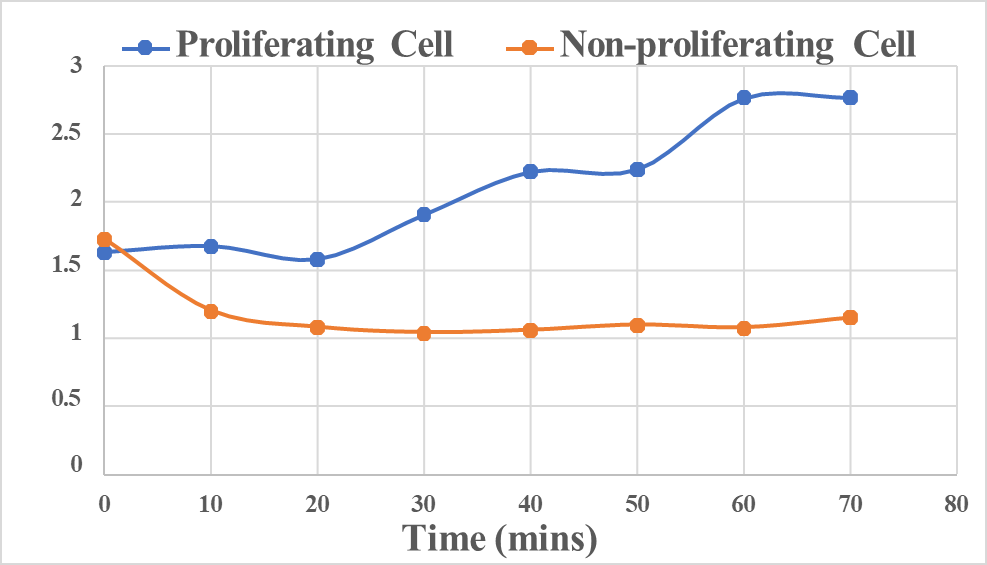
Fluorescence intensity time trajectory for a single proliferating B. subtilis cell and a single non-proliferating B. subtilis cell.
Funding Information
This material is based upon work supported by the U.S. Department of Energy, Office of Science, Office of Biological and Environmental Research (BER), under Award Numbers DE-SC0020338, DE-SC0023184.
